- SAP Community
- Products and Technology
- Enterprise Resource Planning
- ERP Blogs by SAP
- Pharma Cold chain tracking : a prototype
Enterprise Resource Planning Blogs by SAP
Get insights and updates about cloud ERP and RISE with SAP, SAP S/4HANA and SAP S/4HANA Cloud, and more enterprise management capabilities with SAP blog posts.
Turn on suggestions
Auto-suggest helps you quickly narrow down your search results by suggesting possible matches as you type.
Showing results for
former_member10
Participant
Options
- Subscribe to RSS Feed
- Mark as New
- Mark as Read
- Bookmark
- Subscribe
- Printer Friendly Page
- Report Inappropriate Content
11-22-2019
10:39 PM
Introduction
Innovation with the Internet of Things(IoT) has been an on-going effort. Although IoT appeared in the Gartner hype cycle for 2017, it has been around for a very long time and businesses have experimented with this technology since the time we can remember. It's only in the recent past that costs of sensors and IoT devices have reduced significantly encouraging businesses to adopt large scale implementation of IoT use cases taking them into production. Among industries supply chain has most often been the frontrunner in embracing new technological trends and that hasn't been any different in the case of IoT. IoT with its promise to bring better and real-time visibility through the entire supply chain makes for informed decisions which is more proactive than reactive bringing in a vigorous efficiency of a kind never seen before.
Now, with IoT moving past its maturity in the hype cycle, the pharmaceutical industry, not among one to be left behind is rebooting and reinventing itself using this technology. Among many of the use cases, one of the most important use case - Cold chain tracking has seen a lot of advancements in the recent past.
IoT enables automation in cold chain
A cold chain is a temperature-controlled supply chain.There are an increasing number of drugs produced by the pharmaceutical industry which needs to be stored in a strict temperature controlled environment (typically 2 to 8 °C) all along the supply chain at every level right from production to packaging, logistics and delivery. Failing to do so will lead to compromised products unsuitable for consumption and puts patients at risk . Monitoring temperature and time out of refrigeration (TOR) is very critical to ensure product integrity and quality. Bringing IoT closer to cold chain helps in introducing various benefits that we discussed, right from real time monitoring to increased efficiency and transparency.
In this blog I am going to focus on one of the use cases for automation of process and monitoring of Time Out of Refrigeration(TOR) calculation during the manufacturing phase in a cold chain. As the API(Active Pharmaceutical Ingredient) undergoes transformation moving in and out of cold storage through different stages in the final manufacturing phase, products that breach the TOR threshold should be pulled out of the supply chain way before it moves to the next level. Better so, with proactive TOR alerting, it is possible to completely avoid potential excursion by enabling necessary action to mitigate the problem and providing an opportunity to reduce loss and increase profits. As discussed earlier IoT enables this kind of automation by gathering the data needed , generating events, enabling actions and also paving the way for advanced analytics , AI and machine learning.
With this background let's dive into developing the story,
How really a business implements the IoT part of the solution depends on where a business is in terms of their own innovation cycle, their total cost strategy and budget spend on innovation. It is all about tying the physical world to the digital world, and this could be achieved either through simple barcode scanners used to manually record the movement in and out of storage, automating the track and trace of movement in and out of cold storage zones with RFID tags and readers or much better obtaining real time and accurate data using high-performance temperature sensors. SAP supports managing your devices using SAP Leonardo IoT services, while helps you integrate your life sciences and pharmaceutical business processes using SAP ECC or S/4 and SAP ATTP. In this blog lets specifically look at designing the User Experience and prototype.
Note : I worked on prototyping this solution with my team-mates john.astill , kevin.changela along with inputs from industry experts mandar.paralkar , Axel Janssen.
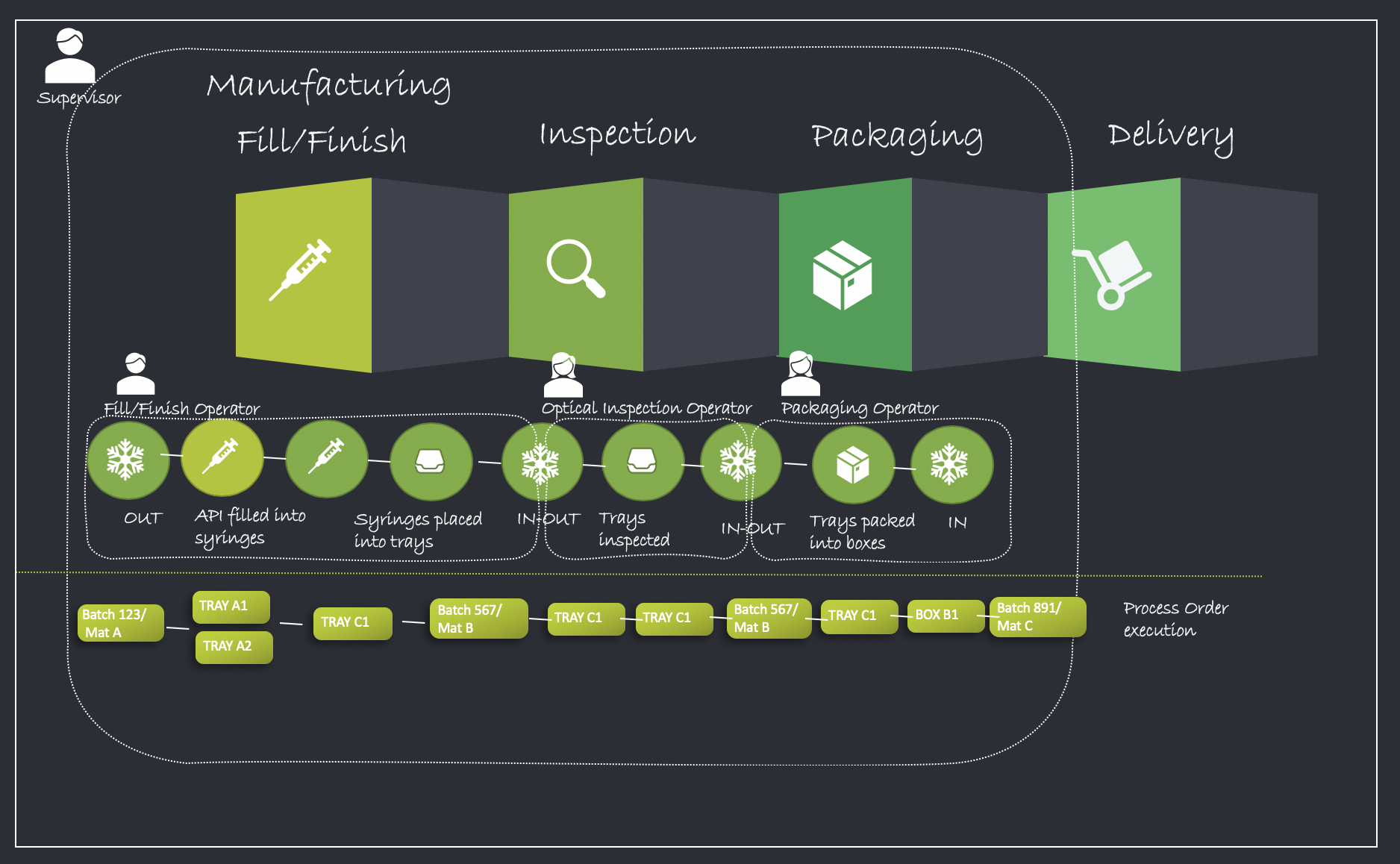
Process Overview
The final manufacturing phase typically consists of 3 stages through which the product moves In/Out of cold storage and undergoes transformation into the final product ready for delivery.
- Fill/Finish
- Inspection
- Packaging
The illustration below details the process flow
Persona :
The persona we will be catering for in the blog is the Supervisor. Let's discover this persona - her goals , motivation, pain points and needs.

User Goal
- Provide real-time insights into the activities on the manufacturing floor for efficient monitoring
- Trigger smart alerts on anomalies to enable proactive decisions/actions.
- Provide detailed view on each production/process order , product transformation details and TOR times for each stage for better traceability.
Assumption
TOR event times like IN_COOL and OUT_COOL is captured for trays/boxes in the backend and association created with the ECC objects like the Process Orders / Batch and Material IDs
User Stories
- As a production supervisor I would like to see all the important information related to the operations on the manufacturing floor in an intuitive and efficient way while allowing me to react to the information quickly.
- As a production supervisor I would like to drill down into specific process orders to view the details for each production step and track the transformation steps and TOR times.
- As a production supervisor I would like to be notified of a potential TOR excursion so that I can take action to ensure the process order is completed within the allowed TOR times.
User Journey
User Journey 1

User Journey 2

User Journey 3
Here's a demo flow with the final prototype:
"As a production supervisor I would like to see all the important information related to the operations on the manufacturing floor in an intuitive and efficient way while allowing me to react to the information quickly."
Ava starts her day by logging in into the Supervisor dashboard built using the Fiori Design guidelines for an Overview Page.

Here, Ava sees all the important information that she needs to know about the current activities on the manufacturing floor. She sees the current status of the Process Orders, alerts, processing times, TOR status etc. calculated real time from the data in the backend. She is also able to view some historical data and the comparison of the current actual measures with the historical averages and defined KPIs. This helps her evaluate the current operational efficiency.
As and when there is any change on the status of the process orders or TOR times (for e.g. completion of the process order/ movement of the process order to the next stage / movement of the product IN/OUT of cold storage) the dashboard is updated immediately.
"As a production supervisor I would like to drill down into specific process orders to view the details for each production step and track the transformation steps and TOR times."
Ava likes to drill down into the process orders and look at the transformation and TOR details at each stage for a process order. Ava would like to get a detailed understanding of the TOR consumption at each stage its association with the trays used, boxes produced at different steps and also their mapping with the batch and material ID in ECC.
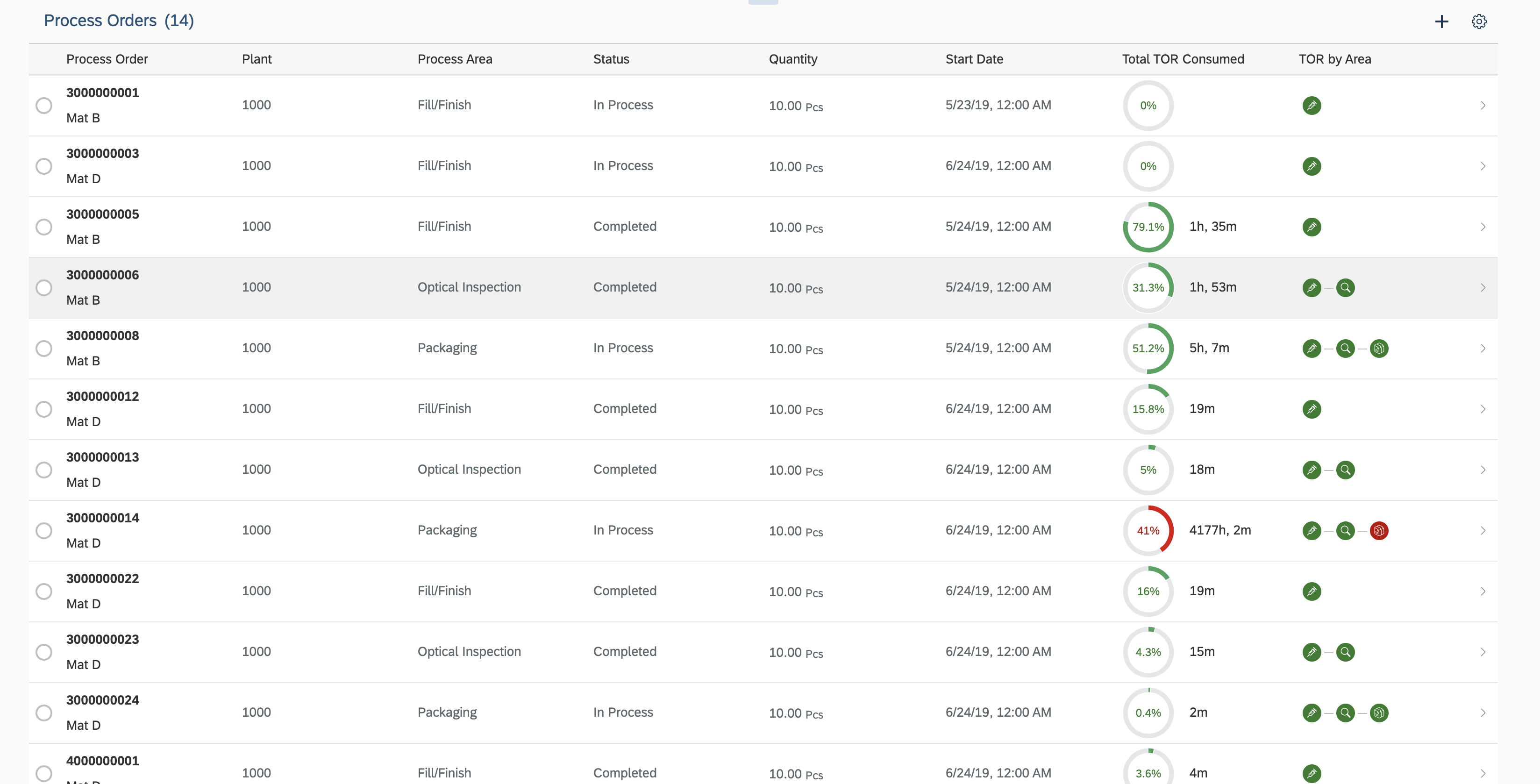
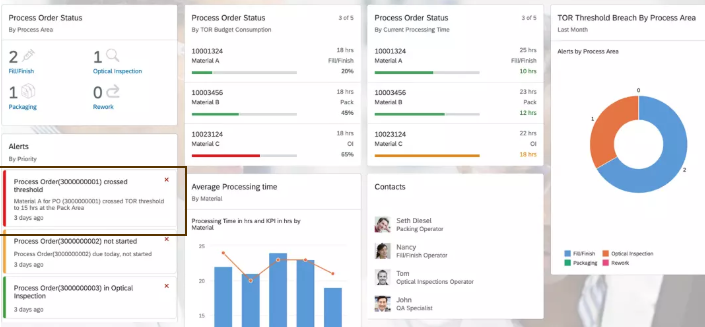
Ava sees a list of the process orders and the high level status on each process order. Ava is able to see which process area/production stage the process order is at, what is the total TOR consumed , whether it is within the TOR limits , has exceeded the threshold or crossed the warning limit for a potential TOR excursion. She can also see whether the TORs have been within limits or not at each stage.
Ava clicks on one of the process order to get a detailed view on the process order.

Ava finds the graphical view of the TOR history extremely useful, as she can get a comprehensive overview on the all the details she needs for that process order. This graphical view chains the transformation events, TOR events and data flow across all the different stages of production. Ava can clearly see which trays were brought out of the cold storage for Fill/Finish , the batch and material IDs associated with the trays and how much time the product was Out of Refrigeration. Ava also sees which trays the transformed product was sent into , associated batch , material id and how much time the product was Out of Refrigeration during Inspection. Lastly she can also see the associated boxes in the packaging stage and TOR times for the packaging stage along with the batch and material ids.
"As a production supervisor I would like to be notified of a potential TOR excursion so that I can take action to ensure the process order is completed within the allowed TOR times."
Ava sees real time alerts on her dashboard for a potential TOR excursion. As she's alerted well in time and also has all the details that she needs to make quick decisions right at her fingertips with the help of this dashboard, she along with the operators are enabled to address the issue and cut losses while ensuring the product is not compromised.
Cheers!
- SAP Managed Tags:
- SAP ERP Central Component,
- Internet of Things
Labels:
1 Comment
You must be a registered user to add a comment. If you've already registered, sign in. Otherwise, register and sign in.
Labels in this area
-
Artificial Intelligence (AI)
1 -
Business Trends
363 -
Business Trends
21 -
Customer COE Basics and Fundamentals
1 -
Digital Transformation with Cloud ERP (DT)
1 -
Event Information
461 -
Event Information
24 -
Expert Insights
114 -
Expert Insights
152 -
General
1 -
Governance and Organization
1 -
Introduction
1 -
Life at SAP
415 -
Life at SAP
2 -
Product Updates
4,685 -
Product Updates
208 -
Roadmap and Strategy
1 -
Technology Updates
1,502 -
Technology Updates
88
Related Content
- SAP S/4 LEAN Integration with Manufacturing Execution systems in Enterprise Resource Planning Blogs by Members
- A web-based interactive 3D Warehouse Monitor from standard data objects in Enterprise Resource Planning Blogs by SAP
- Streamlining Real-time Business Processes with SAP Event Mesh: A Prototype Exploration in Enterprise Resource Planning Blogs by SAP
- SME Partners in Brazil experience the first Hack2Build dedicated to SAP BTP fast adoption for SME use cases in Enterprise Resource Planning Blogs by SAP
- SAP Data Migration Transparency in Enterprise Resource Planning Blogs by SAP
Top kudoed authors
| User | Count |
|---|---|
| 8 | |
| 6 | |
| 5 | |
| 4 | |
| 4 | |
| 3 | |
| 3 | |
| 2 | |
| 2 | |
| 2 |